A new paper from scientists at the Max Planck Institute offers a great look at how HiFi sequencing delivers significantly improved results for metagenome studies compared to short-read data. In this project, HiFi reads led to higher-quality assemblies with less coverage and gave more insight into these complex microbial communities.
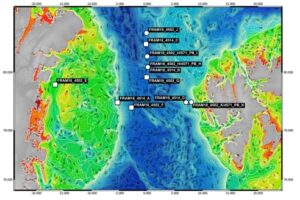
In the PeerJ publication, lead author Taylor Priest (@taylorpriest2), senior author Rudolf Amann, and collaborators report the analysis of 11 seawater samples collected from the Fram Strait, which connects the Arctic and Atlantic oceans and offers a unique view of how climate change is affecting marine ecosystems.
Long-Read Sequencing in Marine Ecosystems Impacted By Climate Change
They performed metagenome sequencing of all samples with short-read technology and analyzed three of them with HiFi reads using the ultra-low library protocol on the Sequel II System. For the PacBio sequencing, all three samples were pooled and sequenced together on a single SMRT Cell, leading to 4-6 Gb of HiFi data per sample.
The PacBio data set yielded 128 metagenome assembled genomes (MAGs) from about 15 Gb of total data collected. In contrast, the short-read data set was about 10 times that size, but produced just 218 MAGs, or fewer than twice as many.
“Of the species-representative MAGs recovered, those generated from the PacBio metagenomes had, on average, larger genome sizes, higher N50 values, and were less fragmented compared to those retrieved from Illumina metagenomes,” Priest et al. report.
The quality of the assemblies also allowed the researchers to simplify their metagenome assembly pipeline. Importantly, the authors note, “taxonomic reassembly was not performed for the PacBio dataset due to the high quality of generated MAGs from single metagenome assemblies.”
HiFi assemblies also showed strength for community composition analysis. In metagenomics, researchers often pull out the 16S rRNA gene sequences to identify the microbial members of a community. Unfortunately, short-read data is not well suited to this task.
“In this study, 84% of MAGs retrieved from the PacBio metagenomes contained at least one complete 16S rRNA gene sequence, highlighting another key advantage of using long Hifi reads.”
This advantage also extended to unassembled data. “A major restriction with short-read metagenomic sequencing is the limited capacity to accurately reassemble full length 16S rRNA genes,” the team notes. “With the advent of highly accurate long read sequences generated from PacBio sequel II (>99% accuracy), full length 16S rRNA genes can be retrieved from single reads without a need for assembly, thus circumventing previous limitations.”
HiFi Reads for Marine Metagenomes Reveals Phylogenetic Diversity
An analysis of these results gave the scientists an interesting look at microbial populations in an area where warming sea water is already having an impact. The recovered diversity encompassed 9 phyla, 11 classes, 27 orders, ∼51 families and ∼54 genera. The most species-rich taxa were the Flavobacteriales (41 species), Pseudomonadales (18 species) and Rhodobacterales (17 species).
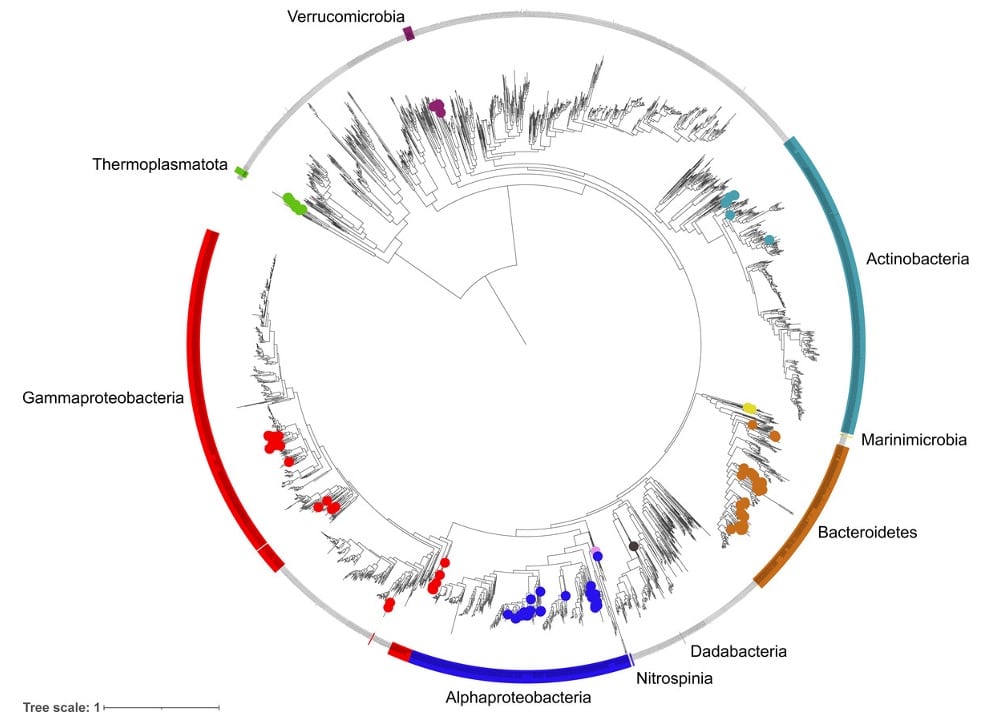
This paper describes the team’s first use of HiFi sequencing for marine metagenomes, but it likely won’t be the last.
“We can conclude that HiFi read metagenomes derived from the PacBio Sequel II platform can greatly improve the number and quality of MAGs recovered, which will allow for further advancement in our understanding of the ecology of marine microbial communities,” they report.
Want to learn more about how HiFi Reads allow researchers to see metagenomes in high resolution? Visit our Microbial Applications page.
Have you heard about our Metagenomics Webinar Series?
Register now for the first episode in our series, highlighting what richer data and better assemblies reveal about metagenome structure and function. Or, stay tuned for the additional webinars in the series showcasing:
● How to resolve viral evolution and quasispecies diversity mechanisms of bacterial virulence and adaptation,
● Identifying key players in host-microbiome interactions with high resolution 16S sequencing, and
● Revealing mechanisms of bacterial virulence and adaptation